The new treatment cures the source of back pain, not just the symptoms.
An injection comprising pulverized vertebral discs has successfully been used to treat degenerative disc disease, one of the world’s most common medical conditions. The therapy was shown to regrow the discs while reducing inflammation and pain, significantly improving the patient’s mobility and quality of life without surgical intervention.
If rolled out to the general population, the team says their treatment could finally alleviate the suffering of millions of chronic lower back pain patients, possibly curbing the rampant opioid epidemic that kills thousands of Americans every year.
The nonsurgical intervention from Summit Medical Center, Oklahoma, tackles chronic low back pain: the most common cause of disability worldwide and the sole cause of more than half of all opioids prescribed in the United States, helping to cripple the global economy.
As there is no gold standard for diagnosing or treating degenerative disc disease, therapies derived from natural materials found in the body called biologics are being explored to regrow these degenerated discs. However, as the disc loses vital liquids as it deteriorates and cracks, to end the pain, the treatment would need to rehydrate these structures as it repopulates native cells to gain the desired height restoration- no easy feat.
In the current study, researchers used a biological platform known as viable disc allograft supplementation, a human tissue graft (allograft) containing micronized or pulverized vertebral discs injected directly into a patient’s damaged disc.
The investigation, involving 50 participants, is part of the larger Viable Allograft Supplemented Disc Regeneration in the Treatment of Patients With Low Back Pain (VAST) Trial, following 220 patients with chronic lower back pain over three years.
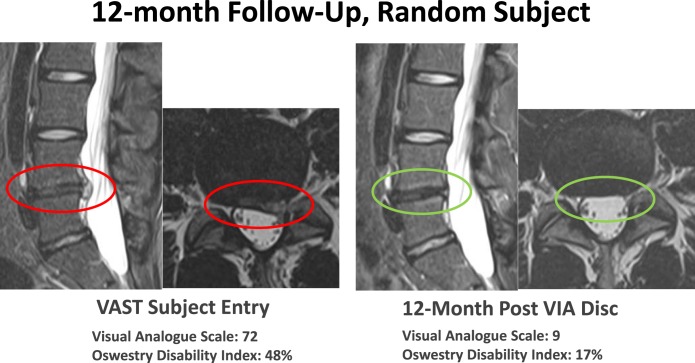
Researchers injected forty-six volunteers with the allograft in this extension, and four received a saline placebo. Using radiography and Magnetic Resonance Imaging (MRI), the scientists saw that the therapy encourages the cells in the damaged discs to regenerate, leaving healthy tissue. These results were also validated using various pain and disability indices which record patient-reported symptoms, functional outcomes, and quality of life. In this way, the group obtained a complete picture of the pain relief gained by the patients, which imaging alone cannot always depict.
The group states the allograft, acquired from cadavers, requires no incisions and can be administered by a clinician using a mild sedative – meaning patients can go home the same day.
“The significant improvement in pain and function is promising for patients living with chronic low back pain – a condition that can greatly impact a person’s quality of life,” said lead author Douglas Beall, chief of radiology at Clinical Radiology of Oklahoma. “Back pain is the leading cause of limited activity and workplace absenteeism. This treatment may help patients return to a normal activity level for a longer period time.”
Why does the back degenerate?
The spine has 26 vertebrae bones cushioned by small gel-filled sacs known as vertebral discs, which act like shock absorbers to allow the flexible movement of the spine. The discs comprise a firm, tough outer layer called the annulus fibrosis, surrounding a soft, jellylike layer, the nucleus pulposus.
Degenerative disc disease occurs when these layers show wear and tear due to aging, injury, or disease. As these structures lose their integrity and dry out, the vertebrae drop to come closer together, reducing the disc’s ability to act like a buttress and provide support. A lack of blood supply means damaged discs do not repair themselves – as a consequence, they can bulge, tear or slip out of place, causing chronic pain and problems with mobility.
The problem with treating this condition is that existing therapies treat the symptoms of the disease rather than the causes.
In the present study, the researchers developed a biologic consisting of a nucleus pulposus allograft, saline components, and a cryopreserved vial of cells (VIA Disc Matrix). According to their past whitepapers, once they thaw the vial, all the ingredients are mixed together and injected using a 22-gauge spinal needle into 1 or 2 degenerated intervertebral discs in the lower spine. To ensure the participants incur no injuries, the scientists use continuous fluoroscopic imaging or computed tomography to guide the procedure, which takes 10-11 minutes for one disc and roughly 15 minutes for two.
Sturdy results in humans
Appearing sound in theory, the researchers have trialed their allograft multiple times. They first ran a one-year pilot study involving 24 patients; next came the larger VAST trial, which followed 220 participants over 12 months; and finally, the newest 50-patient VAST extension study running over three years, whose results the team reported at the Society of Interventional Radiology Annual Scientific Meeting in Phoenix.
In the pilot and large-scale VAST trial, scientists compared the results from participants receiving the allograft to those from the saline placebo and nonsurgical management (NSM) cohorts.
In the pilot study, subjects receiving the allograft exhibited a more considerable reduction of pain and a significant increase in functional improvement than the placebo group over the 12 months. In the 224 patients aged 19 to 73 years, pain improved by fifty-four percent at the one-year mark in patients receiving the allograft – accompanied by a fifty-three percent improvement in the self-reported Oswestry Disability Index (ODI): indicating that disc tissue allograft may be a beneficial nonsurgical treatment for patients who have chronically painful lumbar degenerative discs.
The extension further validates these results by showing that after three years, sixty percent of patients who received the allograft injection for chronic low back pain reported a fifty percent reduction in pain, with seventy percent of this group reporting more than a 20-point improvement in their ODI scores.
Limitations and future plans
Despite the positive results of the studies, the group remains realistic, voicing concerns about the small number of reported adverse events as they lost many participants in the follow-up. And even though these events weren’t linked to the allograft: a larger placebo and NSM group are still needed for comparison to ensure these events are not treatment-related.
For the future, the scientists say further clinical trials are needed to investigate the efficacy of viable disc tissue supplementation using a less-reactive control: as saline has been proven to elicit responses in the nervous system and could affect results.
Despite these limitations, the team says using their disc graft injection could help decrease opioid use among patients with chronic low back pain: “Existing treatment for chronic low back pain due to degenerative disc disease is often ineffective or the effects are short-lived,” said Beall. “We need better treatments for this condition since conservative care is not providing the long-term outcomes that patients deserve. Injectable allograft treatment might be the answer for many people.”

